|
Test attributes are listed in the "Attributes" column. These emergency use authorizations (EUAs) have been issued for each individual test with certain conditions of authorization required of the manufacturer and authorized laboratories. The table below includes information about authorized SARS-CoV-2 molecular diagnostic tests. Individual EUAs for Molecular Diagnostic Tests for SARS-CoV-2 EUA for Molecular Diagnostic Tests for SARS-CoV-2 Developed And Performed By Laboratories Certified Under CLIA To Perform High Complexity Tests.Umbrella EUA for SARS-CoV-2 Molecular Diagnostic Tests for Serial Testing. 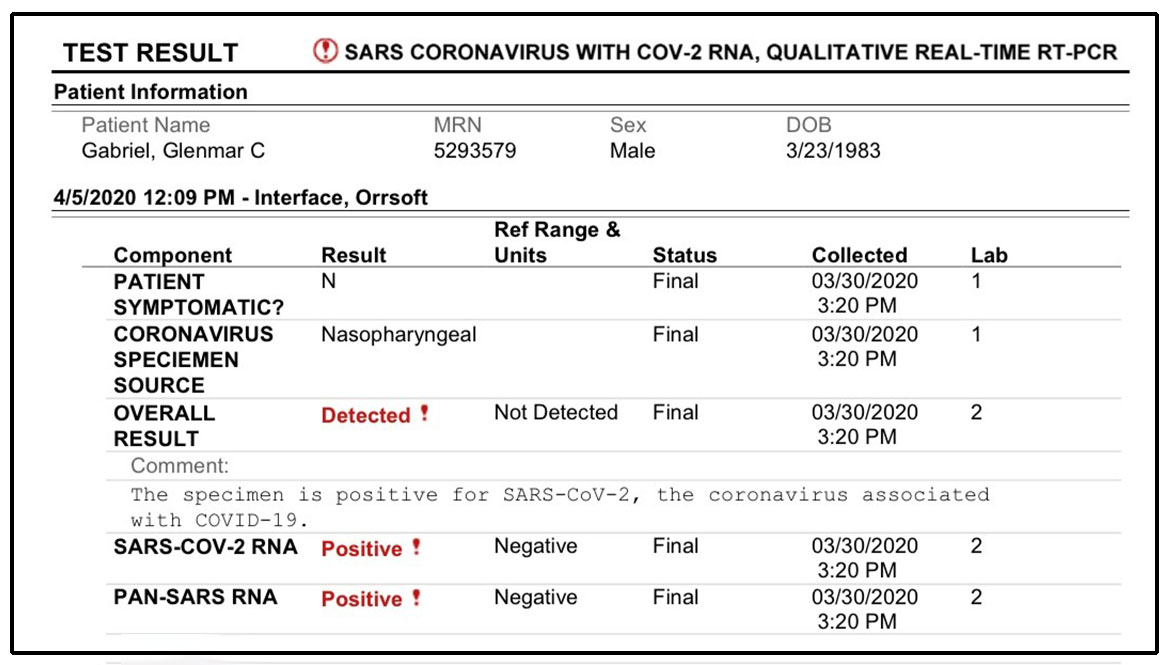
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
